5 Module 5: Microbial Interactions
5.1 Overview
Weeks 8 and 9 focus on co-culture experiments that examine how isolates interact through diffusion-mediated contact.
5.2 Purpose
- Evaluate microbial interactions through co-culture growth of different isolates.
- Observe growth while maintaining fluidic contact across a membrane.
- Analyze possible inhibition or facilitation patterns.
- Use pipettes to seed co-culture wells for growth assays.
- Apply the principles of the Cerillo Duet system to your research question.
5.3 Learning Outcomes
- List safety considerations for propagating microbes.
- Explain the purpose of Duet wells and co-culture assays.
- Discuss the significance of microbial fluid interactions.
- Practice diluting cultures to a target value.
- Seed wells consistently without contamination.
- Describe the purpose, methods, and preliminary results of co-culture experiments.
- Collect and interpret co-culture growth data.
- Revise the draft for the individual and group projects.
- Explain how the experiment contributes to the overall research goal.
5.4 Skills and Knowledge
5.4.1 Skills
- Follow lab safety and PPE protocols.
- Use Cerillo vertical membrane co-culture systems correctly.
- Analyze co-culture growth data.
5.4.2 Knowledge
- PPE requirements for microbial work.
- Co-culture design and growth assessment.
- Microbial interaction concepts.
5.5 Task
Review the background and procedure information before lab and work with your partner to set up the co-culture assays and document results.
5.6 Criteria for Success
Successful completion requires participation in assay setup, collection of analyzable data, and a complete ELN record.
5.7 Background
After characterizing individual growth and metabolism, this module asks how isolates behave in the presence of other organisms. The Cerillo Duet co-culture platform allows isolates to grow in adjacent chambers separated by a membrane that permits exchange of metabolites without direct mixing of cells.
5.8 Procedures
5.8.1 Lab Safety
- Wear required PPE and clean benches and pipettors with 70% ethanol.
- Treat all tips as biohazards.
- Decontaminate liquids, glass, and work surfaces after lab.
5.8.2 Methods
5.8.2.1 What Was Prepared in Advance
- Overnight cultures of each isolate were started in 2.5 mL TSB and incubated at 30 C with shaking.
5.8.2.2 Day of the Lab
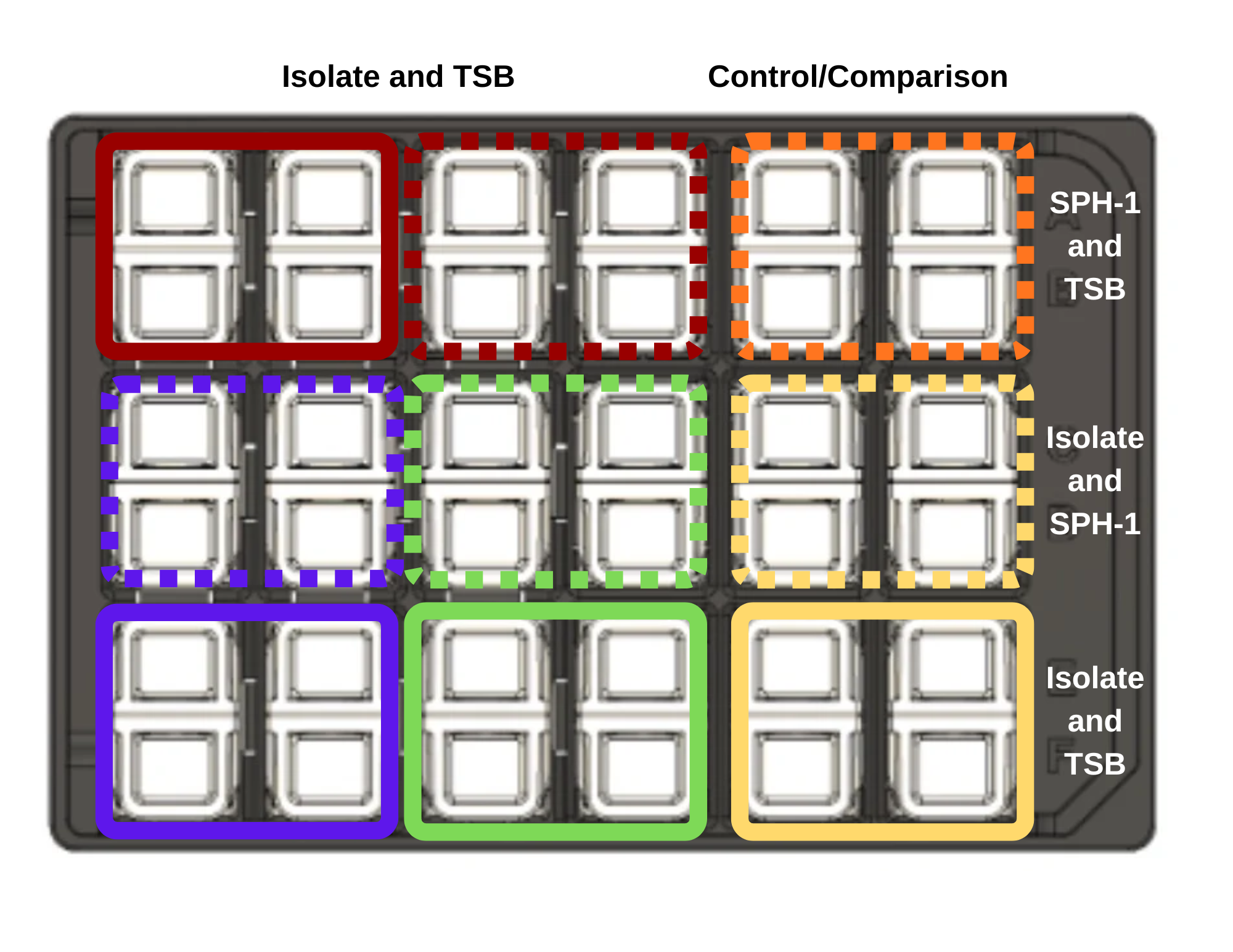
Figure Figure 5.1 maps the isolate-only, control, and co-culture conditions across the Cerillo Duet platform.
- Dilute the overnight culture 1:1000 into fresh TSB.
- Add 800 µL of diluted culture to triplicate wells for the isolate-only condition.
- Add 800 µL of TSB to the adjacent control wells.
- Add 800 µL of diluted SPH-1 to the paired experimental wells for co-culture.
- Seal the plate carefully.
- Incubate with shaking at 30 C for 24 to 48 hours.
- Analyze OD600 measurements collected every 5 minutes.
5.8.3 Protocol Notes
Record any mistakes, deviations, or strain-specific observations.
5.9 Results
Add images of the co-culture plate and any figures generated from the growth data.
5.10 Result Analysis
Compare isolate-only growth to co-culture growth. Explain whether the second organism appeared to support, suppress, or otherwise reshape your isolate’s growth profile.
5.11 Discussion Questions
- How can co-culture systems help researchers understand microbial behavior?
- What kinds of metabolites or resources might explain improved or reduced growth in co-culture?